Leading by Example: SC Johnson's Pioneering step in Corporate Environmental Responsibility
Environmental Leadership as a Model for the Oil Industry
It looks like the end of the world
In 1973, the Mexican chemist Mario Molina joined the laboratory of Professor F. Sherwood (Sherry) Rowland at the University of California, Irvine as a postdoctoral fellow. Rowland was intrigued by James Lovelock’s recent findings that chlorofluorocarbons (CFCs) had spread throughout the atmosphere of both the northern and southern hemispheres. (
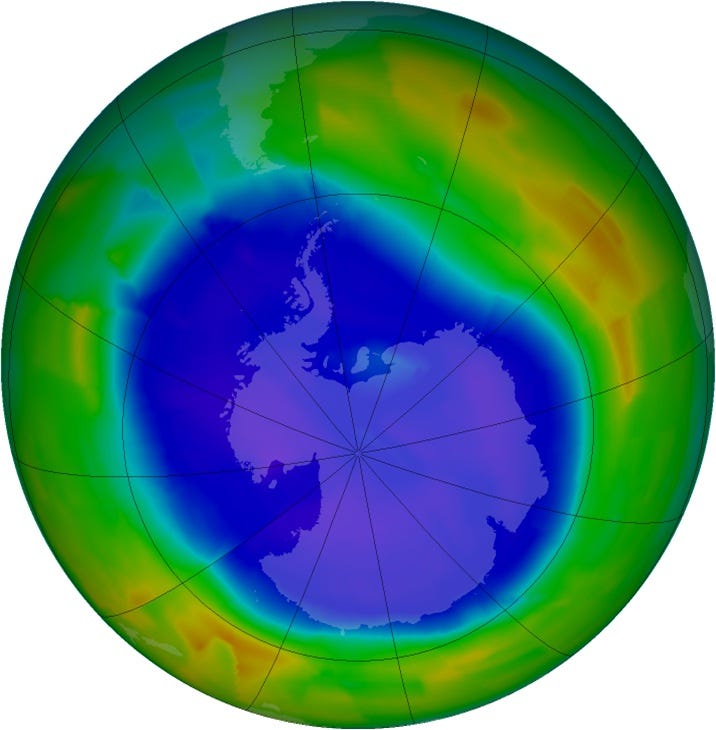
At the time, CFCs were considered harmless and inert compounds. However, Rowland wondered about the potential consequences of releasing them into the atmosphere and suggested that Molina investigate their environmental impacts. Molina’s study, based on computer modelling, soon painted a worrying picture. According to his calculations, CFCs are inert, and they would remain in the atmosphere for a long time—somewhere between 40 and 150 years—before eventually reach the stratosphere. There, they are exposed to ultraviolet radiation (UV) from the sun, which breaks the chemical bonds of the CFC molecules, causing them to release significant amounts of chlorine atoms. These chlorine atoms react with ozone molecules, leading to the destruction of the ozone atmospheric layer.
Ozone is a confusing molecule. At ground level, ozone is a harmful pollutant, and a key component of photochemical smog, a type of air pollution that forms when sunlight interacts with nitrogen oxides (NOx) and volatile organic compounds (VOCs). This smog appears as a brownish or yellow haze and is common in urban areas with heavy traffic and industrial activity. Prolonged exposure can cause respiratory problems, such as asthma and other lung diseases, and can damage, crops, forests, and other vegetation.
In the stratosphere, however, the region of the atmosphere from 6 to 31 miles, ozone absorbs most of the sun's harmful UV radiation and therefore prevents this dangerous radiation from reaching the surface. Without a protective ozone layer in the atmosphere, animals and plants could not exist, at least not on land.
If CFCs could destroy ozone in the stratosphere, the world would be in serious trouble within a few decades. Molina and Rowland realised that their research, which started as an interesting scientific exercise, had turned into a global environmental problem. In their book, "One With Nineveh: Politics, Consumption, and the Human Future", Paul R. Ehrlich and Anne H. Ehrlich, share an anecdote where Rowland after arriving home one evening and being asked about the work by his wife, sombrely replied, “The work is going well, but it looks like the end of the world."
They published their findings in a short paper in Nature magazine, and in September 1974, during the meeting of the American Chemical Society (ACS), they recommended a complete ban on the future release of CFCs into the environment. While the paper in Nature had passed almost unnoticed, a press conference organised by the ACS brought extensive attention to the chlorofluorocarbon-ozone problem.
The Reaction
Their announcements sparked a multifaceted response. While governments, public, and environmental organizations recognised the urgency of addressing the issue, the chemical industry initially reacted with scepticism and opposition, primarily driven by economic interests and the potential regulatory impacts on their operations. CFCs were widely used in refrigeration, air conditioning, and aerosol propellants. Companies, like DuPont, had substantial investments in these products and were concerned that phasing out CFCs would result in significant financial losses and market disruption. Alternatives to CFCs, at the time, were either not developed or considered too expensive. The industry argued that transitioning to new chemicals would require substantial investment in research and retooling of manufacturing processes.
Leading the Change
However, not all companies reacted in the same way. In June 1975, in a groundbreaking move, SC Johnson set a new standard for environmental responsibility by becoming the first major company to eliminate CFCs from its aerosol products.
This move was led by the then-CEO, Sam Johnson, who was convinced by the mounting scientific evidence that CFCs were detrimental to the ozone layer. As he said at the time,
“Our own company scientists confirm that, as a scientific hypothesis, [the idea that fluorocarbon propellants in some aerosol containers might be causing ozone depletion] may be possible. Effective today, our company has removed all fluorocarbon propellants from our production lines.”
This initiative was ahead of its time, taking place over a decade before the Montreal Protocol and several years before the U.S. government mandated the phase-out of CFCs. As it turned out, removing CFCs proved to be the right decision. Twenty years later, the scientific research Sam Johnson relied on to make the decision won the 1995 Nobel Prize.
His decision was good for the business, too. Company scientists found that propane and isobutane, among the cheapest gases, were effective substitutes for CFCs. SC Johnson ultimately saved millions of dollars on aerosol production and was well-established in CFC-free products by the time competitors caught up.
From Aerosols to Oil Rigs
Sam Johnson’s decision was driven by the principle of doing the right thing for the environment, and its customers, regardless of immediate economic impacts. The oil and gas industry, a significant player in the global economy, has a unique opportunity to embrace this ethos of environmental stewardship. Instead of being compelled by impending regulations, the sector can proactively reduce greenhouse gas emissions and adopt cleaner technologies. This proactive approach not only meets global obligations to combat climate change but also promotes innovation, enhances brand reputation, and builds consumer trust. By taking decisive action now, the oil and gas industry can demonstrate its commitment to being part of the solution, rather than contributing to the problem.
If you enjoyed this article and want to support my work, consider buying me a coffee! Your contributions help me continue to provide insightful content on climate history and more. Thank you!
Sources:
https://mediatheque.lindau-nobel.org/laureates/rowland/research-profile
Lovelock , J. E., Maggs, R., & Wade, R. (1973). Halogenated Hydrocarbons in and over the Atlantic. Nature(241), 194-196. doi:https://doi.org/10.1038/241194a0
Molina, M. J., & Rowland, F. S. (1974). Stratospheric sink for chlorofluoromethanes: chlorine atom-catalysed destruction of ozone. Nature, 249(5460), 810-812.
This Week's Citation Classics. (1987, December 7). (49). doi:https://garfield.library.upenn.edu/classics1987/A1987K930400001.pdf
Taking CFCs Out of Aerosols How Sam Johnson Led SC Johnson to Environmental Activism





